The brain’s movement rhythm in ALS
By Alejandra López-Castro
Do you remember Stephen Hawking? The world famous physicist who transformed our understanding of black holes and the universe, was also known for another reason: he lived for decades with amyotrophic lateral sclerosis, or ALS. While Hawking became a symbol of resilience and scientific brilliance, his story also brought global attention to a devastating neurological disease that still has no cure.
ALS is often described as a disease that slowly disconnects the brain from the muscles. Over time, those who suffer the disease progressively lose the ability to move, speak, swallow, and eventually breathe. Yet one of the biggest scientific mysteries is understanding exactly how the disease changes brain activity itself. A recent study published in Aperture Neuro, conducted in the laboratory of Professor Jakob Udby Blicher as part of the PhD research of Tobias Sevelsted Stærmose, explored this question using magnetoencephalography (MEG), a technology capable of detecting the tiny magnetic signals produced by brain cells in real time.
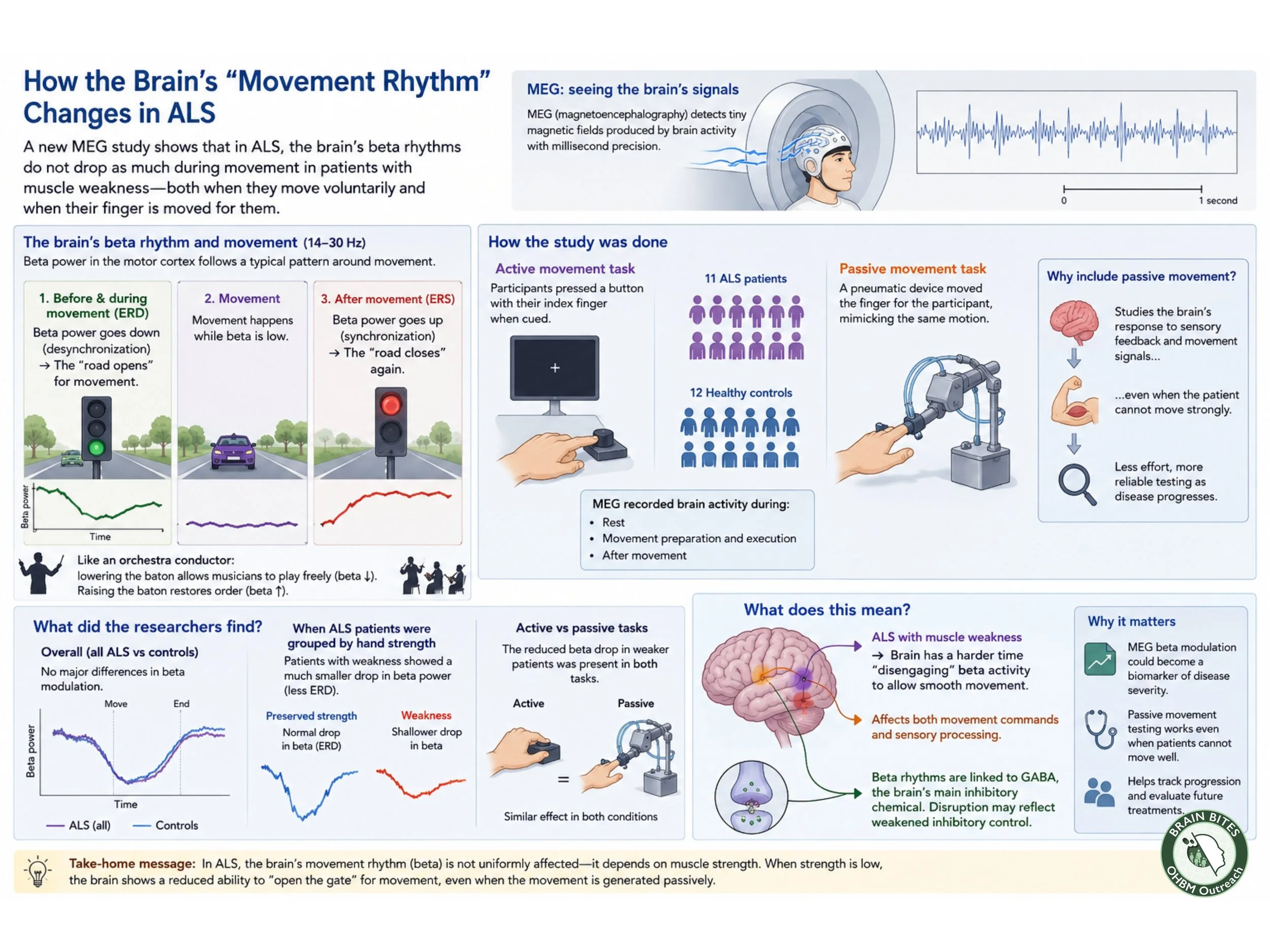
The researcher focused on beta rhythms, a type of brainwave strongly linked to movement and coordination. Normally, these rhythms act almost like a traffic control system in the brain. Before and during movement, beta activity decreases, allowing the brain to open the road for motion. After the movement ends, the rhythm rebounds, restoring order to the motor system. Scientists refer to these phases as event related desynchronization (ERD) and event related synchronization (ERS).
Another way to picture this process is to imagine an orchestra conductor. When the conductor lowers the baton, musicians begin to play more freely and dynamically. In the same way, the brain lowers beta activity to allow smooth movement. Once the action is complete, the conductor restores rhythm and control. In ALS patients with muscle weakness, however, this “conductor” appears less effective, making coordinated movement more difficult.
To study these changes, researchers compared 11 ALS patients with 12 healthy volunteers. Participants either pressed a button with their finger or had their finger moved passively by a pneumatic device designed to mimic the same motion. This passive-movement task was especially important because it allowed scientists to observe brain responses even when patients could not move strongly on their own.
At first, the results seemed subtle. Overall, ALS patients did not show dramatic differences compared with healthy individuals. But when researchers separated patients according to hand strength, a clearer pattern emerged. Patients with significant weakness showed a much smaller drop in beta activity during movement preparation and execution. In simple terms, their brains struggled to switch into movement mode.
This can be compared to driving a car with a delayed accelerator pedal. Pressing the pedal still produces motion, but the response feels slower and weaker. Similarly, the motor system in ALS patients with muscle weakness may have trouble shifting efficiently into the flexible brain state needed for movement.
One of the most interesting findings was that these altered brain rhythms also appeared during passive movement, not only during voluntary actions. This suggests that ALS may affect how the brain processes sensory information from the body, not just how it sends commands to muscles.
The study also points toward a possible biological explanation. Beta rhythms are closely linked to GABA, a neurotransmitter responsible for inhibitory signaling in the brain. Reduced beta modulation may reflect weakened inhibitory control, a process already suspected in ALS.
Importantly, passive movement experiments could become valuable clinical tools in the future. Many neurological tests depend on patients actively performing tasks, which becomes increasingly difficult as ALS progresses. Passive testing, however, is like checking the electrical wiring of a house without needing to turn on every appliance. Even if muscles are too weak to respond normally, researchers can still evaluate how the brain itself is functioning.
Although the study involved a relatively small group of participants, it highlights the potential of MEG as a way to track disease severity and better understand ALS progression. For a disease that still holds many unanswered questions, these brain rhythms may offer scientists a new window into how the nervous system changes long before movement is completely lost.
Sources:
Stærmose TS, Andersen LM, Dalal SS, Bailey CJ, Blicher JU. Movement-related beta modulation in amyotrophic lateral sclerosis depends on muscle strength: A magnetoencephalography study. Aperture Neuro, 2026.
Kiernan MC et al. “Amyotrophic lateral sclerosis.” The Lancet (2011).
Timmins HC, Vucic S, Kiernan MC. “Cortical hyperexcitability in amyotrophic lateral sclerosis.” Current Opinion in Neurology (2023).