A new frame for psychiatry promises an optimistic picture
When I was a trainee in psychiatry, I often had the same conversation with severely ill patients. Frustrated that treatment wasn’t fully helping, they would ask why. “Why do I have this condition? Why, despite taking my medications, don’t I seem to get better? I’ve seen other patients come and go, some even worse than me, and after a few days they seem good as new. But not me. Why?” And I remember a professor of mine always responding the same way. “You want the short answer? We don’t know.”
The long answer to that question is really complicated. The truth is that psychiatry has had significant advances, mainly due to new technologies that have allowed a better understanding of conditions that, for many years, were thought not even to be related to biological mechanisms in the brain. However, the fight is far from over and there is still a lot of ground to cover. We haven’t yet produced a framework that clearly links brain mechanisms to symptoms. Without that link, it’s hard to explain why people respond differently to the same treatment. That’s why Lee and colleagues are proposing a new framework to better approach psychiatric disorders. One that would hopefully translate into not only answering patients’ valid questions but also improving their lives. Dr. Kangjoo Lee, Dr. Jie Lisa Ji, Dr. Markus Helmer, Dr. John D. Murray, Dr. John H. Krystal and Dr. Alan Anticevic have recently published their work in Biological Psychiatry. Here, we will explore a simplified version of the main ideas behind this work (for details, see the original article).
Current limitations in psychiatric research
Over the years, different technologies have allowed us to study the brain. This includes a special modality of Magnetic Resonance Imaging (MRI) called functional MRI (fMRI), which enables researchers to indirectly trace brain activity. Brain activity, as well as its connectivity (how different regions communicate) forms the basis of all behaviour, cognition and personality. The hope was that mapping brain activity and connectivity would improve diagnosis and predict symptom severity, and even predict how patients would respond to treatments. So far, results have been promising but not good enough to change clinical practice. The authors argue that this may be because most studies are identifying patterns in brain data rather than revealing the biological processes that actually drive psychiatric symptoms. It’s like being able to notice the smoke without yet understanding what started the fire.
Without understanding the underlying mechanisms of a disorder, it is extremely difficult to develop biomarkers. Biomarkers are the holy grail for many medical specialties. They are objective measurements, like blood tests or brain scans, that help diagnose illness, predict outcomes, or guide treatment, which Psychiatry still urgently needs.
The authors suggest several changes that could move the field forward. First, focus on mechanisms rather than just symptoms. Second, recognize that multiple mechanisms can produce similar symptoms, and that one mechanism can lead to different symptoms over time. Third, conduct follow-up studies with more than one evaluation. Some psychiatric symptoms may be stable over time but others fluctuate considerably and, ideally, biomarkers should reflect this.
Studying disease mechanisms to understand psychiatric disorders
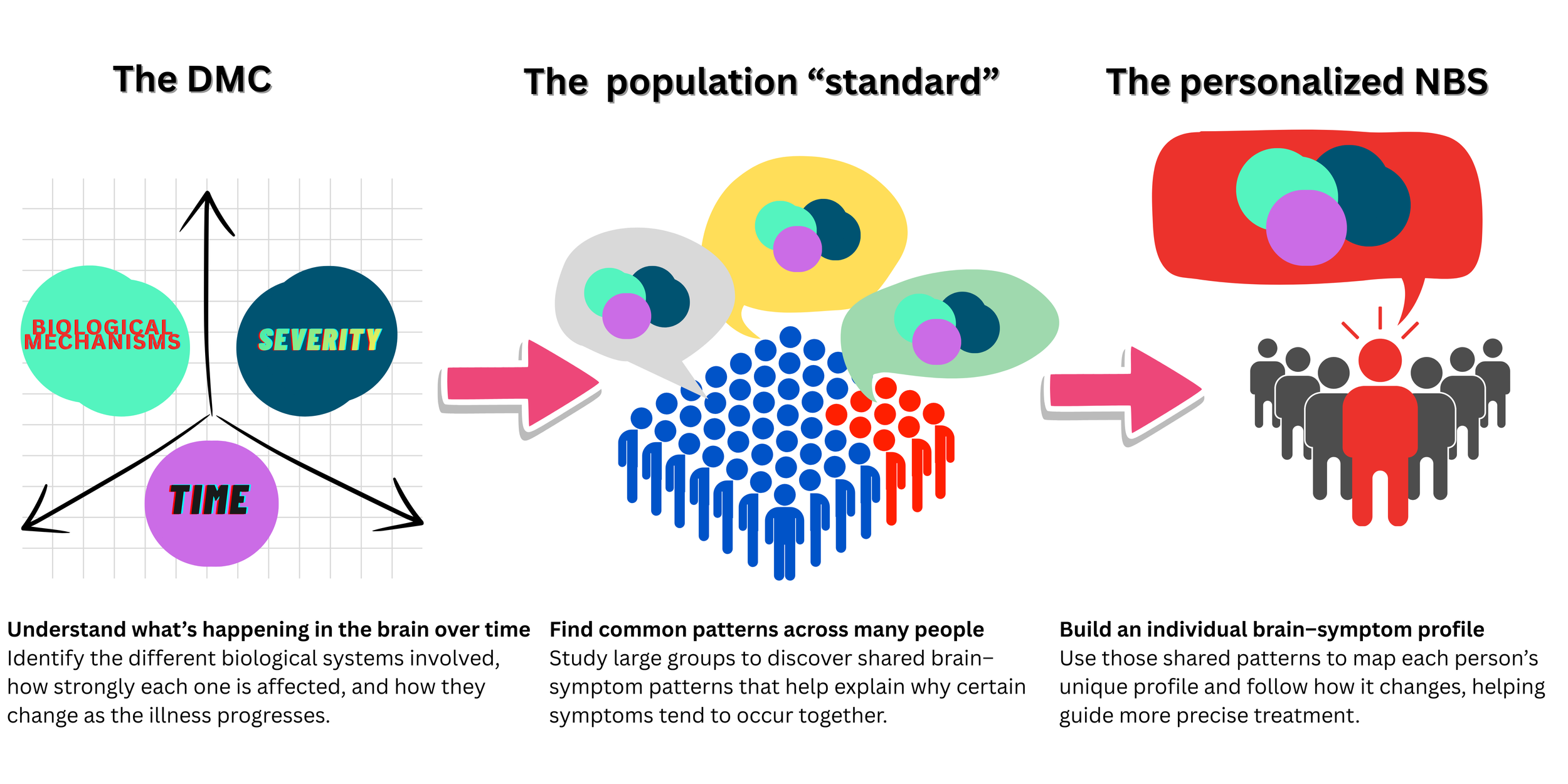
So what exactly does this new framework actually propose? The authors introduce a theoretical model called Dynamic Disease Mechanism Complexity (DMC). A helpful way to understand this is to imagine a sports team, where multiple players interact and influence how the team performs. The DMC has three main components. (1) A Mechanism Domain, which refers to different biological mechanisms, similar to players with distinct roles. (2) Mechanism Severity, which is a measure of how impaired each system is, similar to how well each player is performing. (3) Time, which reflects how mechanisms evolve over the course of an illness, like how a player’s performance can change across games or seasons. Just as a team’s performance depends on all players working together, psychiatric symptoms reflect how multiple biological mechanisms interact at any given time.
For example, in schizophrenia, the mechanism space would include several biological changes associated with the disorder. Key mechanisms linked to schizophrenia include dysregulation of brain chemical systems such as dopamine and glutamate, as well as environmental factors like stress, substance use, and early-life adversity. These mechanisms affect brain circuits to different degrees, which corresponds to mechanism severity. Furthermore, these biological changes can fluctuate depending over time. Dopamine and glutamate-related dysfunction can look different early in the illness compared to years later, and before and after treatment. That is why the third component, time, is equally important. However, if each of these three domains can vary and interact with one another, this means many different combinations are possible.
When we consider all these mechanisms, the complexity quickly becomes overwhelming, something we might expect given the intricacy of the human brain. Researchers therefore need a way to condense this complexity into something more manageable. One way to do this is by reducing thousands of measurements into a few major patterns that capture most of the meaningful variation, much like summarizing a long book into its main themes. Scientists refer to this as a “low-dimensional” subspace, where “space” simply means a way of organizing complex information so we can see patterns more clearly, and “subspace” refers to a simplified part of that larger information map. In this context, the authors propose the concept of Neurobehavioral Subspaces (NBSs), which condense neural features, such as common patterns of brain connectivity, and link them to changes in behavior, like the symptoms observed in patients. Theoretically, researchers could define “reference” spaces that represent the overall pattern of how brain changes relate to symptoms at a population level. They could then construct personalized NBSs, which are an individual’s unique brain-symptom profile over time. This personalized profile could shift with factors like stress, sleep loss, or medication. In simple terms, the mechanistic space explains why the brain is changing, while the NBS shows how those changes relate to symptoms.
Why bother to travel through all these spaces and mathematical models? Because this could be a turning point. This might provide the framework psychiatry desperately needs to make more precise diagnoses, better prognostic and risk predictions and help guide treatment decisions. The work by the authors suggests that this is possible given that this framework and modeling would allow for better prediction, better patient stratification, more precise treatment matching and for testing causal hypotheses. Other specialties in medicine have been advancing at a tremendous rate over recent years. Perhaps the time has come for psychiatry to be the underdog winner of the season.